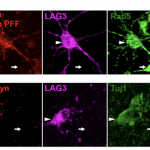
Dr. Gilbert gives an update on stem cell therapies for the treatment of Parkinson’s, including profiling a few notable...see more

Dr. Gilbert gives an update on stem cell therapies for the treatment of Parkinson’s, including profiling a few notable...see more

New York, NY, August 28, 2017 – The American Parkinson Disease Association is pleased to share with the Parkinson’s...see more
Simple Email Signup v2 (Salsa replacement form)