How carbidopa/levodopa is used to treat Parkinson’s disease symptoms
Loss of neurons in the brain that use dopamine to communicate is one of the hallmark features of Parkinson’s disease (PD), causing slowness, stiffness, tremor, and balance problems. Replacing the brain’s dopamine is therefore one of the key treatment strategies to help improve the motor symptoms of PD.

Dopamine itself does not cross the blood-brain barrier and therefore can’t be used to treat PD. Instead, levodopa, a precursor of dopamine, which does cross the blood-brain barrier is used. If levodopa is ingested by itself however, it breaks down in the bloodstream before it crosses into the brain, so levodopa is typically ingested with another medication that stops it from breaking down. In the US, the combination of carbidopa/levodopa is used.
Carbidopa/levodopa is the mainstay of treatment for Parkinson’s disease and is the most effective medication currently available.
APDA research support played a role in the discovery of levodopa for PD treatment when we funded the work of Dr. George C. Cotzias back in the 1960s.
While carbidopa/levodopa remains the most effective medication for people with PD, it is available in various strengths and delivery systems which makes understanding all the available levodopa options very confusing. To help you make sense of the many options, I will describe the various levodopa formulations below, along with the rationale for using each one.
Different formulations and doses of Carbidopa/levodopa for treating Parkinson’s
Carbidopa/levodopa immediate-release (brand name Sinemet) is the original formulation and the one that is most frequently used.
This is typically the first formulation to be tried when someone is started on levodopa treatment. This medication comes in three dosages 10/100, 25/100 and 25/250. The first number refers to the milligrams of carbidopa in the pill. The second number refers to the milligrams of levodopa in the pill.
Early on in PD, a dose can last six hours or longer, but often, as PD progresses, a levodopa dose tends to relieve symptoms for shorter and shorter amounts of time. A person may take a dose of levodopa and have good symptom relief for a period of time, referred to as ON time. Then the effects of levodopa may start to wear off and symptoms of PD return for a period of time, referred to as OFF time.
This phenomenon of alternating between ON and OFF time is known as motor fluctuations. There are many strategies that your doctor can try to smooth out your response to medication throughout the day and these are summarized in a recent APDA webinar.
If this is happening, one strategy is to try a different formulation of levodopa that is designed to last longer than immediate-release carbidopa/levodopa. Another strategy is to try a formulation of levodopa that can be used as needed if the medication effect wears off between oral doses of medication.
Carbidopa/levodopa formulations that may last longer than Carbidopa/levodopa immediate-release
Carbidopa/levodopa extended-release (brand name Sinemet CR)
This is a formulation of carbidopa/levodopa that was originally designed to be extended or controlled release. For some people it works well as an option to extend the life of a dose of carbidopa/levodopa. Sometimes, this medication is prescribed at bedtime to help with nighttime symptoms and to try to prevent OFF time in the morning. The brand-name version of this medication is no longer being manufactured, although a number of generics are available. The dosages of this medication are 25/100 and 50/200. Again, the first number refers to the milligrams of carbidopa in the pill and the second number refers to the milligrams of levodopa in the pill.
Despite its “extended-release” formulation, for many people this formulation does not last longer than carbidopa/levodopa immediate-release. Other formulations were therefore designed to achieve this goal.
(Carbidopa/levodopa/entacapone (brand name Stalevo®)
This is a combination medication of carbidopa/levodopa and entacapone, designed to last longer than carbidopa/levodopa alone. Entacapone is a COMT (cathechol-O-methyltransferase) inhibitor which, in a manner similar to carbidopa, inhibits the breakdown of levodopa in the periphery and allows more levodopa to cross the blood brain barrier and get converted into dopamine in the brain.
This medication comes in a number of dosages as follows:
| carbidopa | levodopa | entacapone | |
| Stalevo 50 | 12.5 mg | 50 mg | 200 mg |
| Stalevo 75 | 18.75 mg | 75 mg | 200 mg |
| Stalevo 100 | 25 mg | 100 mg | 200 mg |
| Stalevo 125 | 31.25 mg | 125 mg | 200 mg |
| Stalevo 150 | 37.5 mg | 150 mg | 200 mg |
| Stalevo 200 | 50 mg | 200 mg | 200 mg |
Carbidopa/levodopa extended-release capsules (brand name Rytary®)
In this formulation of carbidopa/levodopa, varying types of beads are packaged within a capsule. The different beads are designed to release the levodopa at different rates, allowing for a more prolonged release of the medication from the stomach. Four strengths are available: 23.75/95, 36.25/145, 48.75/195, 61.25/245 (with the first number again referring to the milligrams of carbidopa in the pill and the second number refers to the milligrams of levodopa in the pill.) A typical dose of medication is three capsules, three to four times a day. Capsules can be opened, and the beads can be added to food without losing their extended-release properties. This can be useful for someone with swallowing difficulties.
Carbidopa/levodopa extended-release capsules (brand name Crexont®)
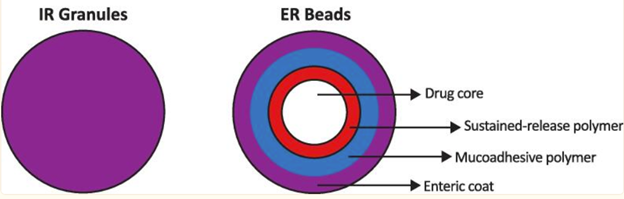
Approved in 2024, the Crexont formulation of carbidopa/levodopa has beads of different types within a capsule, with the immediate-release pellets dissolving first and extended-release granules dissolving more slowly. The difference between Rytary and Crexont is that Crexont beads have additional features that allow for a longer time over which the medication is released and absorbed.
- There is an enteric coating on the outside of the granules that prevents the beads from disintegrating too early in the stomach
- Once the enteric coating is off and the beads make their way to the small intestine for absorption, there is a mucoadhesive polymer that keep the beads adhered to the area of absorption for longer
- In addition, there is a coat of a sustained-release polymer to enable slow release of the drug once the drug is at the small intestine
Source LeWitt, P et al doi: 10.1016/j.prdoa.2023.100197.
Four strengths are available 35/140, 52.5/210, 70/280, 87.5/350. A typical dose is 1-2 capsules, 2-4 times a day depending on levodopa requirements.
Carbidopa/levodopa enteral suspension (brand name DuopaTM)
Instead of taking carbidopa/levodopa in a pill form, people with PD can receive carbidopa/levodopa in a gel form infused directly into the small intestine where levodopa is known to be absorbed. This system can be useful for those with advancing PD who have motor fluctuations that are no longer controlled by oral medications alone. The system can be particularly helpful for those who have gastroparesis, or delayed gastric emptying, which is a common non-motor symptom of PD and can keep oral medications stuck in the stomach and therefore unable to be absorbed readily by the small intestine.
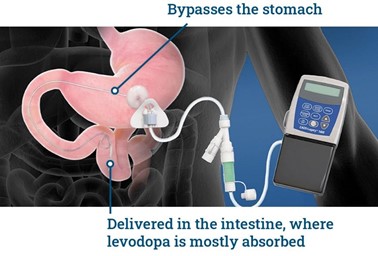
This is how the system works (diagram above). In a minor surgical procedure, a gastroenterologist inserts a PEG/J (percutaneous endoscopic gastrostomy with jejunal) tube with one end in the small intestine and the other end emerging from a stoma or hole in the abdominal wall. The tubing that emerges from the abdomen is then attached to a small portable pump, which can be worn in a carrying case over the shoulder or around the waist. The pump is in turn connected to a cassette of carbidopa/levodopa gel.
After the system is inserted, the person with PD works with his/her neurologist to find the right settings for the pump.
There are three doses of medication (also called a bolus) that need to be optimized:
| Morning bolus | The amount of levodopa that is infused into the small intestine when the system is started in the morning This bolus allows the person to turn “ON” in the morning |
| Infusion rate | The rate at which the gel is infused into the small intestine over the course of 16 hours of the day |
| As-needed bolus | The amount of levodopa that is infused into the small intestine on an “as needed” basis when the pump is activated by the person with PD Used for unexpected OFF times during the day |
Your doctor will show you how to properly care for the stoma and explain any potential issues to be mindful of.
For someone with Duopa in place, the daily routine may be as follows:
- A set of carbidopa/levodopa enteral suspension cassettes (each containing 2,000 mg of levodopa – a person with high levodopa requirements may need two cassettes over the course of a day) are stored in the refrigerator
- In the morning, a cassette is removed from the refrigerator 20 minutes prior to use and attached to the pump once it reaches room temperature
- The cassette tubing is attached to the tubing that emerges from the abdomen
- The pump is turned on and activated to give the morning bolus
- Then the pump automatically starts delivering the gel at the predetermined continuous rate
- The pump can be inserted into its carrying case so that it is portable as the person with PD performs their daily activities
- The person with PD can activate the pump to deliver pre-programmed extra doses throughout the day if necessary
- At the end of the day, the pump is removed from the carrying case and shut off
- The cassette tubing and the abdominal tubing are disconnected. The tubing that emerges from the abdomen is flushed with saline and capped for the night
- If medication is needed during the night, the person may be given oral doses of carbidopa/levodopa to take
Continuous subcutaneous carbidopa/levodopa infusion (brand name VyalevTM)
Approved in 2024, VyalevTM is a subcutaneous 24-hour infusion of foslevodopa/foscarbidopa for the treatment of motor fluctuations in Parkinson’s disease (PD). Foslevodopa/foscarbidopa is a soluble form of carbidopa/levodopa which is quickly converted into carbidopa/levodopa and can lead to steady blood levels of levodopa within 24 hours.
Carbidopa/levodopa formulations to be used as needed
Carbidopa/levodopa orally disintegrating tablets (brand name Parcopa®)
This formulation of carbidopa/levodopa dissolves in the mouth. Although it is absorbed in the gut, the fact that it does not have to be broken down in the stomach means that onset of action of the medication is somewhat quicker than regular Sinemet. It is also useful for those who have difficulty swallowing pills.
Levodopa inhalation powder (brand name Inbrija®)
This is an inhalation formulation (containing only levodopa) designed for quicker onset of action, to be used as needed if medication effects wear off between oral doses of carbidopa-levodopa. The dose is taken when PD symptoms start to return. Absorption of the medication takes place through the lungs which bypass absorption through the gastrointestinal tract. When a person is experiencing return of his/her symptoms, he or she breaks a blister pack containing a 42 mg levodopa capsule and inserts it into the Inbrija inhaler device and inhales through the device. Then a second capsule is inserted into the device and inhaled (one dose is two capsules). This can be done one time during each OFF period, up to five times a day. The inhaler cannot be preloaded with the capsules which must stay in their blister package until immediately before use. Only an Inbrija inhaler can be used with the levodopa capsules.
Tips and Takeaways
- Carbidopa/levodopa is the mainstay of PD treatment medications
- Carbidopa/levodopa comes in a variety of formulations and doses and seeks to replenish the dopamine in the brain that is lost due to PD
- Some formulations help to smooth out motor fluctuations
- Other formulations are used “as needed” if PD symptoms return between doses
- Consult your neurologist if your current dosage is no longer working as effectively as before, and/or to determine whether there is a new formulation that may benefit you